 
This allows for the implanted stimulation leads to remain internalized during the trial. These anchored stimulation leads are then connected to temporary extension leads, which are externalized. Ī second method of trialing stimulation therapy is referred to as a “staged” or “buried lead trial.” In this procedure, an incision is made at the time of trial initiation and the leads are anchored to the paraspinal fascia. The permanent leads are reintroduced to the same location in the epidural space, anchored to the paraspinal fascia via a small incision, and tunneled to a generator implanted in the flank, gluteal area, or abdominal wall. Our practice is to wait until the needle holes from trial have healed (7–10 days), to avoid operating near tissues with a preexisting wound. There are no formal guidelines on how long to wait before permanent device implantation after a trial. 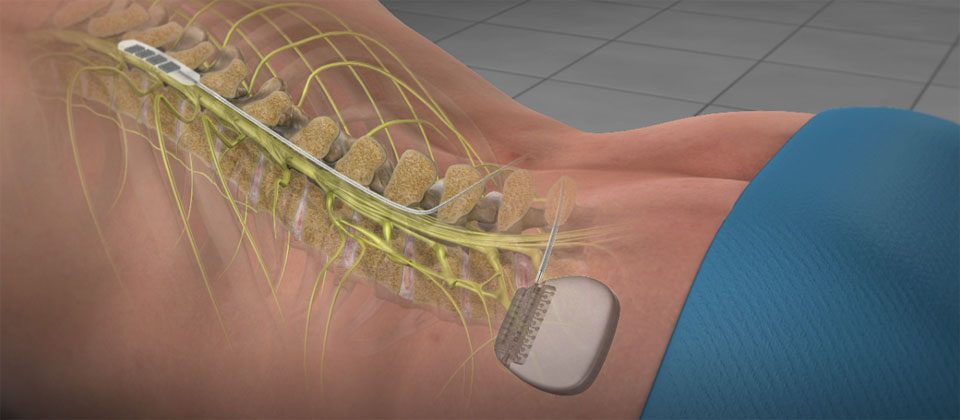
If the trial phase is satisfactory, a permanent device is implanted several days or weeks later. The temporary leads are removed at the conclusion of the therapeutic trial. The patient is allowed to experience stimulation therapy for several days to assess their response. During the trial period, temporary leads are inserted into the epidural space percutaneously and connected to an external pulse generator. To assess the patient’s response to therapy, a trial phase is usually conducted prior to implantation of a permanent device. Ĭomponents of the spinal cord stimulator. SCS devices have 2 main types of leads: surgical paddle leads, which require a laminotomy, and cylindrical leads that are placed percutaneously. The SCS device consists of an implantable pulse generator connected to 1 or more stimulating leads introduced into the epidural space and positioned at the level where the pain is best inhibited ( Figure 1). The purpose of this review is to summarize the epidemiology and pathogenesis of SCS infections, provide guidance for diagnostic evaluation and management, and discuss strategies for prevention. SCS infection has significant implications for the device recipient, often requiring device removal, preventing patients from receiving adequate pain management and adding to the initial cost and disability. However, mortality associated with SCS implantation is infrequent. Nearly 50% of these complications warrant subsequent surgical revisions. 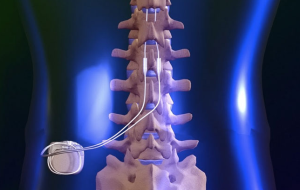
SCS implantation is not without complications, with an overall estimated rate up to 40%. The number of SCS implantations is steadily increasing, and the individual cost associated with these procedures can reach up to $60 000. 
Indications for SCS use include neuropathic pain, intractable angina, cancer-related pain, complex regional pain syndrome, and painful peripheral arterial disease. SCS consists of implantation of electrodes in the epidural space to alter painful stimuli, originating in the dorsal columns of the spinal cord, to the brain. Patients who fail conservative treatments may be considered for electrical neuromodulation therapy, among which spinal cord stimulation (SCS) is the most commonly utilized technology. Several pharmacological and nonpharmacological therapies are available for management of chronic pain. Spinal cord stimulator, infection, diagnosis, management, prevention Several preventive strategies can be incorporated in surgical practice to reduce the risk of SCS infection. Duration of antimicrobial therapy depends on severity of clinical presentation and presence or absence of associated complications. While superficial surgical site infection following SCS implant may be treated with antibiotic therapy alone, deep infection involving implant warrants device removal to achieve cure. Early recognition of SCS-related infections and associated complications is based on clinical suspicion, laboratory testing, and appropriate diagnostic imaging. However, delay in diagnosis may lead to complications such as meningitis, epidural abscess, and/or vertebral osteomyelitis. Most SCS infections present as generator pocket infection. Infection is one of the most dreaded complications related to SCS implantation and may prevent patients from receiving adequate pain treatment, adding to the initial cost and disability. Spinal cord stimulation (SCS) is the most utilized invasive electrical neuromodulation treatment for the management of refractory chronic pain syndromes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
